
Monitoring cancer cells effectively can help physicians with treatment and management, thus reducing cancer-related mortality. Can non-invasive technologies pave the way for improved monitoring to reduce cancer mortality rates? Diagnostic platforms that non-invasively measure the electrical properties of cancer cells offer promise in the early detection of cancer drug resistance and metastasis.
Research has shown that it is possible to understand a cancer type and its drug resistance status from cellular permittivity and conductivity data. In fact, there is an increasing demand for analytical methods that can rapidly measure a cell’s electrical properties.
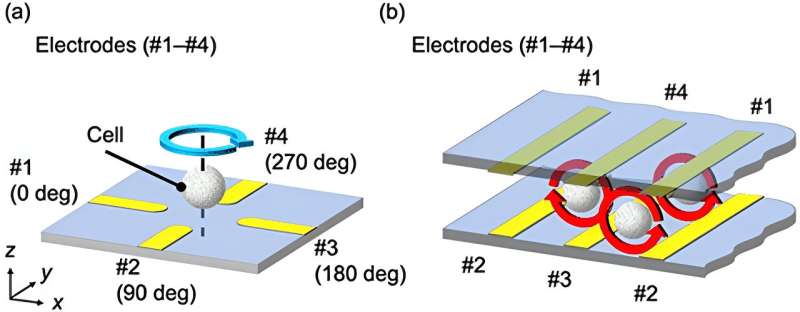
Electrorotation (ROT) offers one such route to capture cellular properties by inferring permittivity and conductivity from a cell’s movement in an electric field. This allows the characterization of the cell type and state by profiling its frequency-dependent rotational movement under a modulated electric field.
However, there are limitations. The challenge is that the capture, measurement, and replacement of cells is quite cumbersome and lowers the throughput of ROT platforms, where throughput refers to the number of cells that a given technology can analyze at any given time.
Recently, researchers from Tokyo University of Science (TUS) developed a continuous flow ROT (cROT) to address conventional ROT’s drawbacks. The new platform leverages microfluidics to continuously measure cellular dynamics and simultaneously capture cells to collect measurements on one device. The group’s validated findings were recently published in Lab on a Chip.
“I discovered that cancer cells had vastly different responses to electric fields while they looked similar. This implied a certain degree of individuality, and the idea of discerning the differences using ROT intrigued me,” explains Dr. Masahiro Motosuke, a Professor in the Department of Mechanical Engineering at TUS and the project’s Principal Investigator. “However, gathering accurate data using ROT requires the precise placement and removal of a single cell, and I wanted to make the process of analyzing many cells easier.”
The researchers fabricated the new device with redesigned interdigitating electrodes that induce cell rotation and a microchannel for cell passage. The electrode geometry increases the number of cells that can be analyzed and reduces the time required to replace a cell as measurements are collected.
The electrical field applied within the microchannel enables analyzing rotational behavior from a continuous flow of cells. Together, these improvements increase the automated system’s throughput. The research team validated the system’s accuracy by obtaining cell membrane permittivity and cytoplasm conductivity measurements from HeLa cells, a human cell line commonly used in research.

