The US Food and Drug Administration has just given the green light to a groundbreaking medical treatment that employs Crispr gene editing.
Known as Casgevy, this innovative therapy is set to help patients with sickle cell disease, an inherited blood disorder that affects over 100,000 individuals in the United States. The UK had previously approved this revolutionary treatment back on November 16. The treatment is a joint effort between Vertex Pharmaceuticals of Boston and Crispr Therapeutics of Switzerland.
This therapy’s primary goal is to alleviate the excruciating pain episodes that come with sickle cell disease. It involves editing the patient’s cells outside the body to counteract the genetic defect responsible for the disease. Eligible recipients will include adults, as well as children 12 and older, who suffer from frequent pain attacks. The therapy is designed to be administered only once, with potential benefits lasting for years, or possibly even decades.
Vence Bonham, the acting deputy director of the National Human Genome Research Institute at the National Institutes of Health, states, “This is a major milestone. This could truly change the lives of individuals and reduce the burden of pain episodes.”
The FDA will make a decision by March 30th on whether the therapy should be approved for beta thalassemia, a related blood disorder. The UK’s November approval included both sickle cell and beta thalassemia.
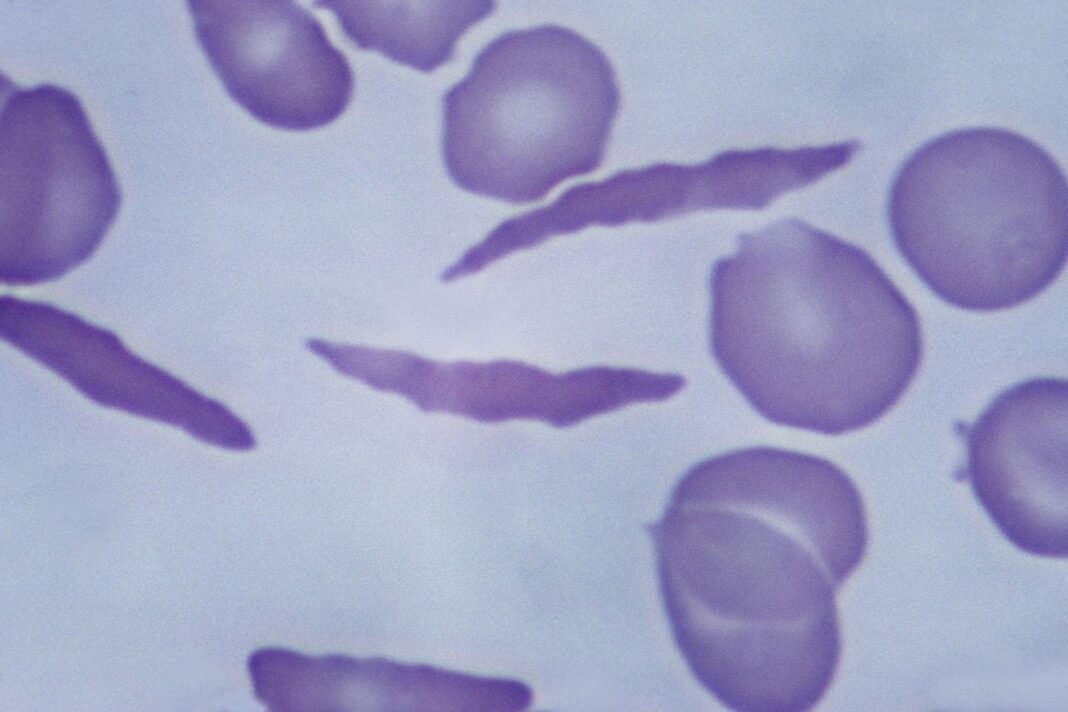
Sickle cell disease causes a person’s red blood cells to be hard and crescent-shaped instead of flexible and round, leading to clumping and blocking of blood flow, resulting in bouts of extreme pain and organ damage. The disease also causes a shortage of healthy red blood cells, leading to anemia, which results in fatigue, breathlessness, and dizziness. People with the disease have a life expectancy that’s more than two decades shorter than the rest of the US population.
Samarth Kulkarni, president and CEO of Crispr Therapeutics, describes the disease as “terrible,” stating that “every day feels like a big burden. Patients have this constant specter of mortality hanging over them.”
The root of the problem lies in abnormal hemoglobin, the protein responsible for carrying oxygen through the body. This arises from a mutation in the HBB gene, wherein children born with sickle cell disease inherit a copy of the mutated gene from both parents.
Casgevy employs Crispr, a Nobel Prize–winning technology, to modify patients’ cells to produce healthy hemoglobin. With Crispr consisting of a protein that cuts genetic material and a guide molecule that directs it where to make the cut, a patient’s stem cells are taken out of their bone marrow and edited in a laboratory. This involves making a single cut in a different gene, called BCL11A, to activate the production of a fetal form of hemoglobin that typically turns off shortly after birth.