
Now presenting an eco-friendly and innovative approach for the blue light-promoted synthesis of heterocyclic thiochromenopyrroledione derivatives catalyzed by titanium dioxide. Credit goes to Professor Yutaka Hitomi from Doshisha University.
Heterocyclic compounds are organic molecules with a ring structure comprising at least two or more elements. In most cases, these rings are composed of carbon atoms along with one or more other elements such as nitrogen, oxygen, or sulfur. They are highly sought after as raw materials in the chemical and pharmaceutical industry, owing to their versatility and excellent physiological activities.
While several methods are available for synthesizing these compounds, most of them involve high temperature and pressure conditions, or the use of precious metal catalysts, adding to the economic and environmental cost of producing heterocyclic organic compounds.
A simple yet effective method for overcoming these challenges has been proposed by a team of researchers from Japan and Bangladesh. Their study was recently published in the journal Advanced Synthesis & Catalysis. Using the proposed strategy, the team demonstrated the synthesis of 20 sulfur-containing heterocyclic compounds in the presence of photocatalyst titanium dioxide (TiO2) and visible light.
Lead by Professor Yutaka Hitomi from Doshisha University, the study was co-authored by a Ph.D. candidate Pijush Kanti Roy from the same university, Associate Professor Sayuri Okunaka from Tokyo City University, and Dr. Hiromasa Tokudome from Research Institute, TOTO Ltd.
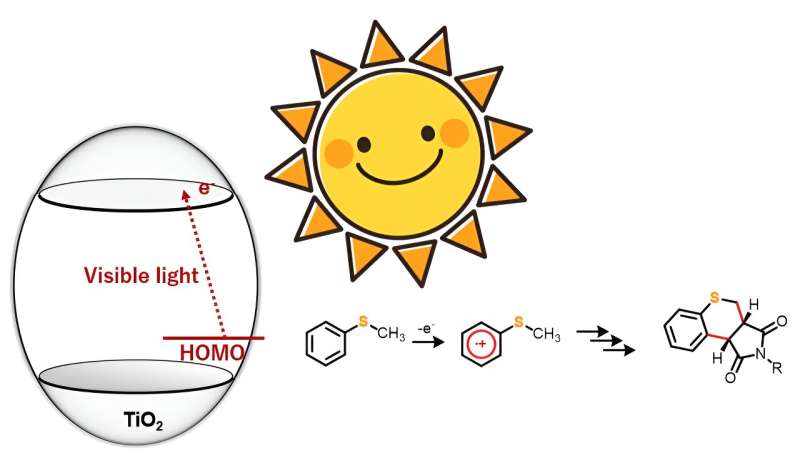
Interest in TiO2 as a photocatalyst for driving organic reactions has been growing among synthetic chemists. The research team observed that under anaerobic conditions, sulfur-containing organic compounds like thioanisole derivatives, when hit with blue light, reacted with maleimide derivatives to form dual carbon–carbon bonds, yielding a new heterocyclic organic compound.
“Unlike ultraviolet light, which generates highly oxidative holes, our approach allows for the selective one-electron oxidation of the substrate molecules using visible light. This approach can thus be employed in various organic chemical reactions,” explains Prof. Hitomi.
The researchers chose five 4-substituted thioanisoles and four N-substituted maleimides for the annulation or ring formation reactions. The team irradiated the starting material with blue light (wavelength > 420 nm) but observed no reaction. However, introducing TiO2 into the reaction system led to the synthesis of 20 different thiochromenopyrroledione derivatives with moderate-to-high yield. They found that within 12 hours of exposure to blue light, the reaction between thioanisole and N-benzylmaleimide led to the formation of a thiochromenopyrroledione derivative with 43% yield, which was close to the theoretical maximum yield of 50%.
The research team also observed substituent effect in the reactions to understand the corresponding mechanistic aspects. From the results, they postulated that the reaction proceeds through charge transfer from thioanisole to the conduction band of TiO2. Furthermore, they suggested that irradiation with blue light triggered one-electron oxidation of thioanisole,

